 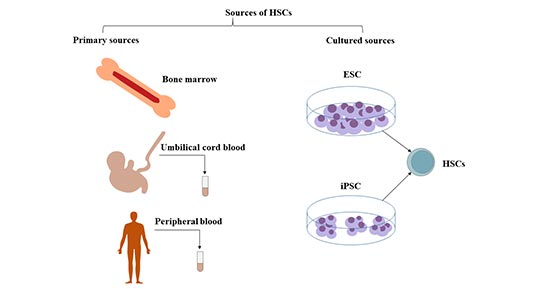 
Bulk cell analysis assumes that each cell, which has the same phenotype, possesses an identical function. 1).Īlthough the classical model has been very useful for understanding the differentiation process of HSCs, it is worth noting that this model has some shortcomings in that it oversimplifies the complexity of hematopoietic stem and progenitor cells (HSPCs), and it is only based on the surface markers and transplantation using bulk cells. All these populations form a tree-like and balanced hierarchy model, within which key transcription factors (TFs) and cytokines precisely conduct the stepwise differentiation of HSCs to mature blood cells (Zhu and Emerson, 2002 Robb, 2007 Metcalf, 2008 Zhang and Lodish, 2008 Seita and Weissman, 2010) (Fig. CLPs further form T, B, NK and dendritic cells, while GMPs differentiate into granulocytes/monocytes and MEPs generate megakaryocytes/erythrocytes. The second branch point at CMPs segregates bipotent granulocyte-macrophage (GMPs) and megakaryocyte-erythrocyte progenitors (MEPs). The first bifurcation occurs between the common myeloid progenitors (CMPs, with myeloid, erythroid and megakaryocytic potential) and common lymphoid progenitors (CLPs, with only lymphoid potential), which are derived from MPPs. LT-HSCs differentiate into ST-HSCs, and subsequently, ST-HSCs differentiate into multipotent progenitors (MPPs), which have no detectable self-renewal ability (Yang et al., 2005). LT-HSCs are a rare, quiescent population in bone marrow and have full long-term (> 3~4 months) reconstitution capacity, whereas ST-HSCs only have a short-term (mostly < 1 month) reconstitution ability. In this classical model, HSCs can be divided into two subpopulations according to their CD34 expression: CD34 − long-term (LT)-HSCs and CD34 + short-term (ST)-HSCs. To better illustrate the relationship between an HSC and its progenies, and the stepwise differentiation process, the immunophenotype-based tree-like hierarchy model was largely established by Weissman’s group (Kondo et al., 1997 Morrison et al., 1997 Akashi et al., 2000 Manz et al., 2002). By contrast, progenitors have been defined by the absence of self-renewal and restricted lineage differentiation capacities. Through transplantation and colony assay, HSCs have been defined on the basis of two essential properties, self-renewal and multipotent differentiation, which can produce cells of all blood lineages (Morrison et al., 1995 Orkin, 2000 Reya et al., 2001 Dick, 2003 Reya, 2003). Since similar approaches can be used to identify multi- and unipotent progenitors, different progenitor populations were also isolated based on surface markers (Kondo et al., 1997 Akashi et al., 2000 Adolfsson et al., 2005 Wilson et al., 2008 Pietras et al., 2015). are still commonly used to isolate HSCs in different labs (Ikuta and Weissman, 1992 Okada et al., 1992 Osawa et al., 1996 Kiel et al., 2005 Oguro et al., 2013). To date, CD34, Sca-1, c-Kit, the signaling lymphocyte activation molecule (SLAM) markers, etc. Since then, different groups have put great effort into identifying more surface markers to further purify HSCs. 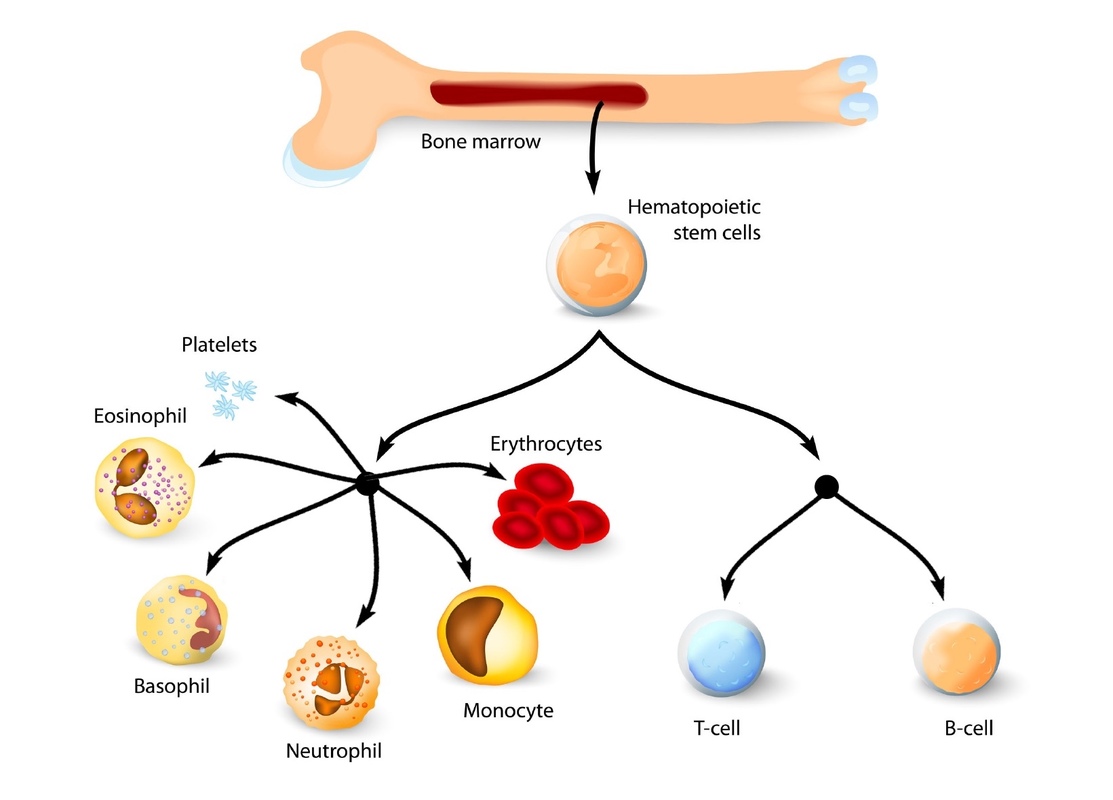
Weissman and colleagues first described HSC-enriched cells using the combination of several surface markers in 1988 (Spangrude et al., 1988). Separation of HSCs became possible with the utilization of antibodies and fluorescence-activated cell sorting (FACS). Thereafter, scientists were interested in developing methods to purify HSCs from bone marrow to better understand their function and molecular regulatory networks. The first in vivo evidence for the existence of HSCs, in 1961, was based on the rescue of lethally irradiated recipient mice by bone marrow transplantation, followed by observing hematopoietic colonies in the spleens of recipients (Till and Mc, 1961). 
This assay has long been the gold-standard for functional HSCs. The cellular potential of hematopoietic stem cells (HSCs) has been traditionally defined by transplanting donor cells (or a single cell) into recipients that are preconditioned by lethal irradiation and therefore devoid of a functional endogenous hematopoietic system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
